Case 1: Motor Vehicle Accident with Entrapment
Evolving Shock and Damage Control Resuscitation in Complex Multisystem Trauma
The Dispatch
EMS Agency Information: Professional Fire-EMS, Paramedic-staffed ambulances and fire apparatus, High-Volume Suburban-to-Rural
Dispatch: Medic 1, Rescue 1 respond to a 29-Delta-3, motor vehicle accident with high velocity impact.
It is a summer afternoon, mid-70s, with light cloud cover, at approximately 1300 hours. Dispatch informs units that multiple callers reported a sedan traveling at high speed in and out of traffic before colliding with the rear of a loaded dump truck parked at a construction site. The incident is in one of the more rural areas of the district along a highway off-ramp. Witnesses on scene report a single male driver entrapped and groaning. No other patients. While units are en route, dispatch updates them: a BLS interfacility transport (IFT) ambulance on scene - not involved in the incident - is reporting that the driver is entrapped and not responding. The call is upgraded to a 29-Delta-7 (MVC with unconscious patient) and an EMS battalion chief equipped with blood products is dispatched from the next nearest station house approximately 15 minutes from the scene.
Closest Level 1 Trauma Center: Approximately 30 minutes by ground with favorable driving conditions, 15 minutes by air with favorable flight conditions
Closest Emergency Department: 13 minutes by ground (Level 3 Trauma Center)
Relevant Geography: Mostly flat highway roads, mild-to-moderate traffic
The Call
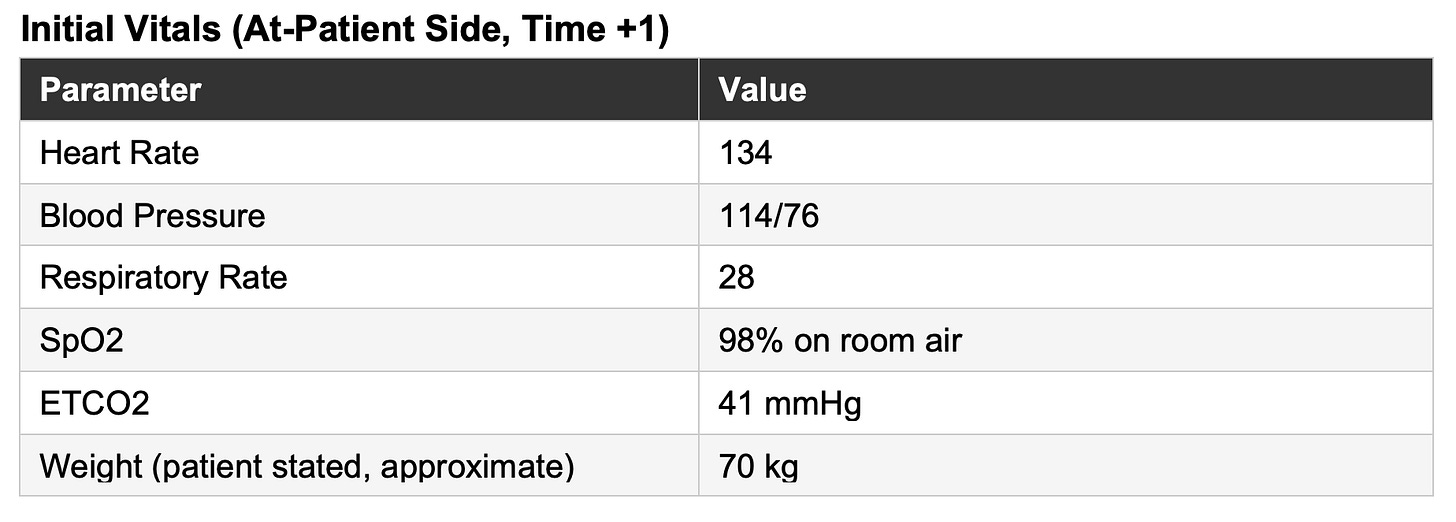
Arriving crews find a 34-year-old male restrained driver in a 4-door sedan with severe front-end damage - the engine block has been displaced rearward and is sitting on the patient’s hips, the dashboard rolled up against his pelvis. He is accessible from the waist up with no chest restriction and has symmetric chest rise bilaterally. The BLS IFT crew has applied a cervical collar; they report a heart rate of 130, a fingerstick blood sugar of 110, and that the patient was unresponsive for approximately two minutes before regaining consciousness.
A paramedic enters the rear compartment passenger side to begin assessment while extrication is set up. The patient is awake and yelling in significant pain. Crews notice that he is mildly pale and clammy with a strong odor of alcohol on his breath.
The patient is awake, alert, and oriented but slow to respond - frequently yelling loudly in pain. He states he is intoxicated, confirms the brief loss of consciousness, and reports head, chest, hip, and leg pain. He states he was going 100 mph.
He has a hypertension on losartan and alcohol use disorder. He denies anticoagulants and antiplatelet agents, and denies known drug allergies.
The limited physical exam is notable for a seatbelt-line bruise across the anterior chest wall with clear lung sounds bilaterally, and diffuse pelvic tenderness with crepitus.
An 18-gauge IV is established in the right antecubital fossa. The crew starts a 1-liter lactated Ringer’s bolus and gives 70 mcg of fentanyl IV.
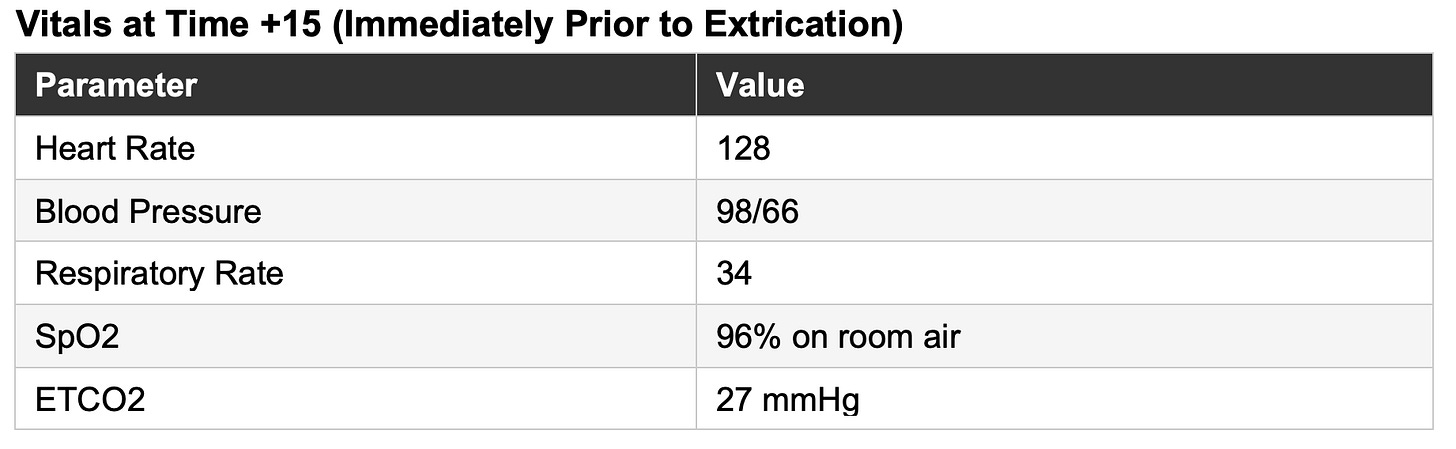
Extrication takes approximately 12 minutes. The patient is removed using a Kendrick Extrication Device (KED) applied to secure and splint the pelvis, as explicitly noted by the crew in the narrative. He is somnolent but easily rousable by voice after fentanyl and reports improved pain.
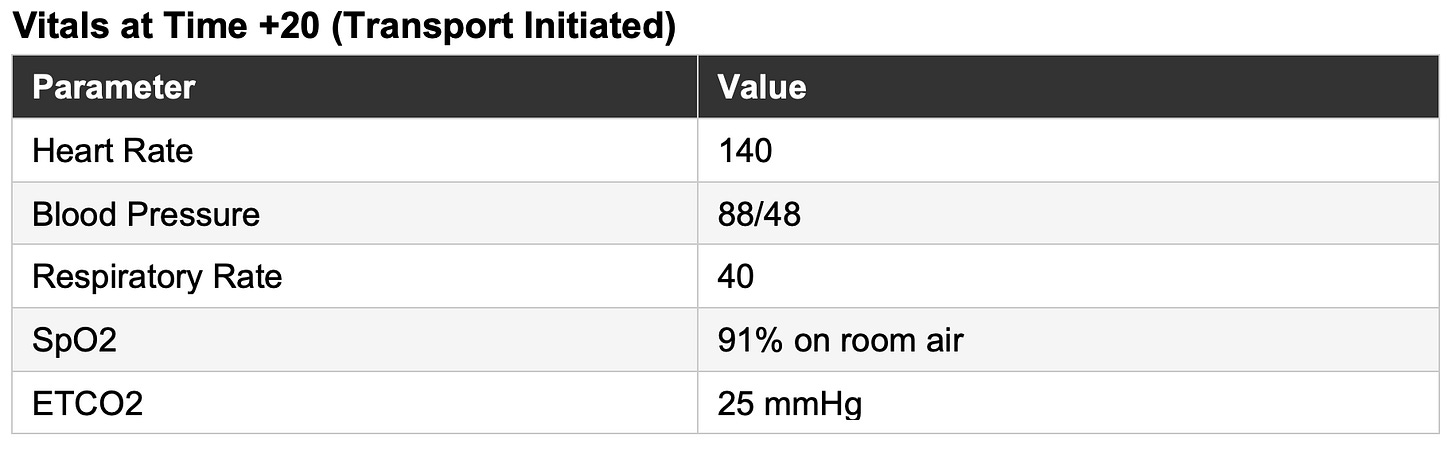
The patient is moved to the stretcher and loaded supine. At time +20, as ground transport is initiated to the Level 1 trauma center, the EMS battalion chief arrives and joins the crew for transport, giving a total of three transporting ALS EMS clinicians.
Shortly thereafter, the patient becomes rapidly more pale, diaphoretic, and lethargic. Radial pulses are thready. He is placed on 15 liters per minute of oxygen non-rebreather. The EMS crew places a 16-gauge IV in the left antecubital fossa and initiates 1 unit of warmed packed red blood cells. On repeat auscultation, he’s found to have absent right-sided breath sounds without JVD or tracheal deviation.
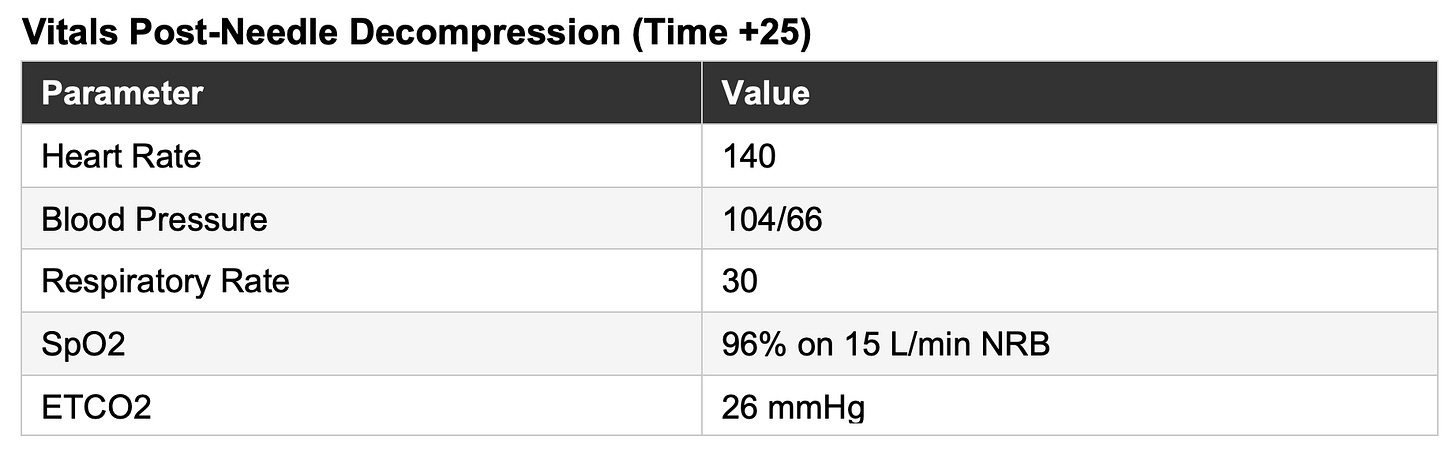
A commercial 14-gauge needle decompression is performed at the 4th intercostal space, anterior midaxillary line, with an audible rush of air. He receives tranexamic acid (TXA).
Secondary Exam - Pertinent Findings:
Diffuse right anterior chest wall ecchymosis with crepitus
Bilateral pelvic crepitus
Right proximal leg crepitus with deformity
Right distal tibia with mangled appearance and patchy oozing lacerations concerning for open fracture; no distal motor or sensation; DP/PT pulses not palpable due to deformity/swelling, but capillary refill 2-3 seconds at the great toe.
Distal pulse, motor, and sensation intact in all other extremities
The KED is removed and a commercial pelvic binder is applied at time +27. The patient remains awake throughout transport, though his blood pressure continues to trend downward. He arrives at the trauma bay awake, with a blood pressure of 94/50 and heart rate of 142.
Emergency Department and Labs
In the ED, he receives additional blood products and a right-sided chest tube for pneumothorax confirmed on ultrasound.
Notable lab work: hemoglobin 9 g/dL, lactate 8 mmol/L, pH 7.15, ethanol level 200 mg/dL.
Notable imaging findings: right-sided rib fractures with pulmonary contusions and a resolving pneumothorax post-chest tube; multiple pelvic fractures with a posterior presacral venous plexus bleed; a right proximal femoral fracture; and multiple right distal tibia fractures.
Hospital Course
The patient underwent posterior presacral venous plexus embolization and surgical repair of his extremity fractures and pelvic injuries. His hospitalization was complicated by acute alcohol withdrawal and a complex surgical site infection at the right distal tibia wounds, ultimately requiring below-knee amputation. After a total hospitalization of seven weeks, he was discharged neurologically intact to a subacute rehabilitation facility.
Overall Reflection
This was a genuinely challenging call, and the crew performed at an extremely high level; a strong example of what high-performing EMS looks like in practice.
Several themes stand out on my review:
The crew recognized shock early and continued to reassess. An initial blood pressure of 114/76 could have been falsely reassuring; it was not! The tachycardia, pale and clammy skin, evolving mental status, and high-energy mechanism kept hemorrhagic shock at the forefront of their assessment and management. This was particularly important in a patient whose intoxication, and later fentanyl administration, could easily have been used to explain away a fluctuating level of consciousness.
Pain control was addressed early. A yelling, entrapped trauma patient cannot be adequately assessed. The fentanyl dose was reasonable.
Evolving hemorrhagic shock was recognized and blood product and TXA were administered rapidly once available. While I would personally avoid large-volume crystalloid in this context, the isotonic crystalloid administration reflects a common bridge practice in systems where blood is not immediately available prehospital.
The tension pneumothorax was clinically diagnosed thanks to a high-index of suspicion and serial reassessments, and treated rapidly during transport. Needle decompression was performed at the correct anatomic landmark with appropriate equipment.
Pelvic stabilization was applied as soon as extrication allowed. The KED provided some compression during the extrication itself, and a commercial binder followed shortly after the patient reached the stretcher.
Finally, the crew made the right transport decisions. What may appear simple in hindsight — bypassing a closer facility and choosing ground over air — is often complex and nuanced in real time.
Now let’s slow this call down and dig into the data behind these learning themes - unpacking the (patho)physiology, the evidence, and the different perspectives that shape clinical decision-making in the field.
Focused Clinical Analysis and Commentary
1. Shock Recognition: Dynamic Assessment and Shock Index Trajectory
Early recognition of hemorrhagic shock is one of the most consequential determinants of trauma outcomes. Traditional reliance on isolated blood pressure values is insufficient; particularly in young, mostly healthy patients with intact compensatory mechanisms who can maintain a near-normal systolic pressure despite significant hemorrhage. In this case, the initial blood pressure of 114/76 could have narrowed the cognitive aperture around shock; the heart rate of 134 should have kept it wide open.
Shock index; defined as heart rate divided by systolic blood pressure; often outperforms either value alone in predicting transfusion requirement, massive transfusion activation, and mortality in trauma, with moderate accuracy across large data sets (Cannon et al., 2009; Carsetti et al., 2023). Another way to say that is, shock index performs meaningfully better than heart rate or blood pressure alone when trying to predict which trauma patients are developing badness.
An SI above 1 is associated with increased blood transfusion need; values at or above 1.4 are classified as severe shock and carry significantly elevated mortality risk. It’s important to mention that SI utility is most reliable in patients without major comorbidities such as hypertension, beta-blocker use, or diabetes, which can alter the tachycardiac response (Carsetti et al., 2023).
Although much of the literature uses transfusion requirement as the primary outcome, need for transfusion is fundamentally a marker of severity. In that sense, shock index functions as a practical, field-ready surrogate for a more immediate clinical question in the field: How sick is this patient right now?
More important than any individual value of the shock index in the prehospital environment, is the trajectory. Trending SI across sequential vitals sets, combined with serial mental status assessment, skin perfusion findings (capillary refill, skin color/pallor), and ETCO₂ monitoring, provides the most complete prehospital picture of shock available without laboratory data.
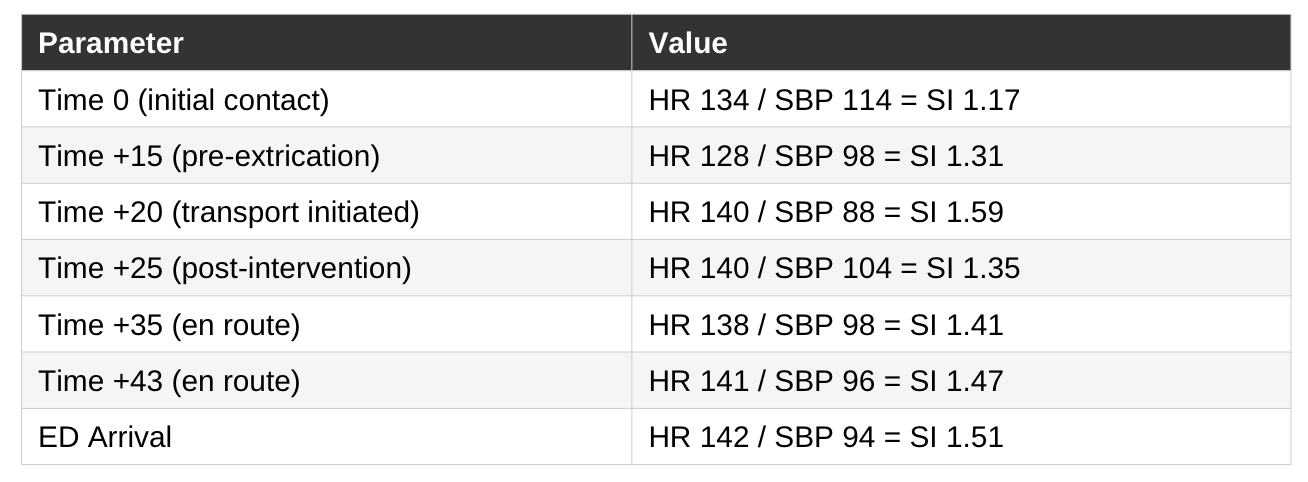
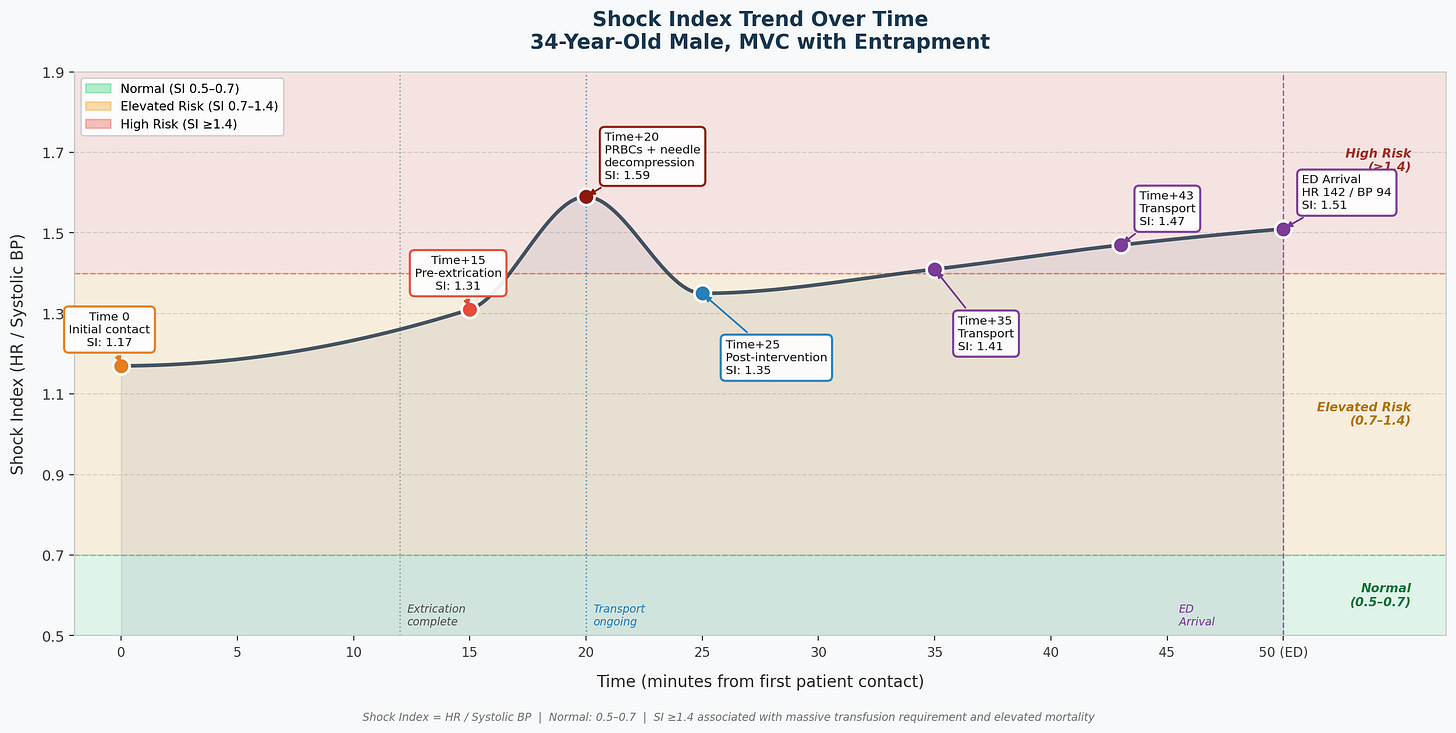
Let’s take a look at the trended SI’s in this patient, as an example:
The trend becomes even clearer when visualized; what appears as a series of isolated vital signs at the bedside becomes a story of subtle decline (rising SI).
Most experienced clinicians can take one look at a patient like this and know: he needed the OR and he needed blood. That part wasn’t subtle. But the real learning pearl here is what the trended SI gives you in the patient who isn’t so obvious - the one whose pressure looks okay, whose deterioration is slow enough to miss.
Likewise, serial ETCO₂ monitoring further supported the evolving shock state. In a spontaneously breathing patient, declining perfusion and worsening metabolic acidosis stimulate compensatory hyperventilation, producing a falling ETCO₂. The reduction from 41 mmHg to 25 mmHg over 20 minutes is consistent with decompensating shock. Prehospital data in trauma patients support ETCO₂ values below 25 to 30 mmHg as markers of severe shock physiology, massive transfusion need, and worse outcomes (Campion et al., 2020; Campion et al., 2022; Bryant et al., 2021).
My recommendation, and my personal approach, is to implement a structured “shock assessment bundle”. While not a formally validated composite score, it a clinical “trick” of mine that integrates complementary and easily accessible data points. My shock assessment bundle: Shock index, ETCO₂, mental status, and skin perfusion (capillary refill, skin color/pallor) together form a dynamic framework that enables prehospital clinicians to detect early deterioration and stay ahead of decompensation rather than reacting to it.
Everyone may develop their own variation, but the key is having a consistent mechanism that translates clinical gestalt into objective data you can track.
2. Prehospital Fluid Strategy in Suspected Hemorrhage
The decision to administer a 1-liter lactated Ringer’s bolus immediately after IV access was established raises an important question: what clinical framework was driving that choice? Was it prophylactic in anticipation of treating a crush-related injury? Was it administered before extrication under the assumption that release of vascular compression would precipitate hemodynamic collapse? Both are plausible explanations.
Isotonic crystalloid as a bridge therapy remains common practice in systems without immediate access to blood products, and in that context it is not inherently unreasonable. It may also be appropriate in the setting of true concern for rapidly evolving crush syndrome, where early volume administration can mitigate rhabdomyolysis-associated renal injury.
The concern in this case is not crystalloid itself; it is the 1-liter bolus delivered in a patient with suspected acute hemorrhage.
Crystalloid does not carry oxygen, does not correct coagulopathy, and in large volumes contributes to dilutional effects that may worsen trauma-induced coagulopathy. When blood is unavailable, the ideal approach is small aliquots; 250 to 500 mL; titrated toward a permissive hypotension target rather than a liter at a time (McMullan et al., 2024).
The 2025 NAEMSP Prehospital Trauma Compendium recommends blood components over crystalloids as first-line therapy for life-threatening traumatic hemorrhage in systems capable of supporting a prehospital transfusion program (Brown et al., 2025). The NAEMSP fluid resuscitation position statement similarly emphasizes limiting large-volume crystalloid and endorses permissive hypotension in patients without suspected traumatic brain injury; targeting an SBP of 90 to 100 mmHg (McMullan et al., 2024).
Traumatic brain injury was on the differential from the moment loss of consciousness was confirmed following a high-energy mechanism in an intoxicated patient with head pain. In damage control resuscitation, permissive hypotension is contraindicated in suspected TBI; secondary cerebral ischemia from even brief hypotension independently worsens neurologic outcomes. Accordingly, the Prehospital Blood Transfusion Coalition clinical practice guideline recommends maintaining SBP ≥110 mmHg when TBI is suspected (Levy et al., 2025).
Both hemorrhagic shock and TBI were being considered in this patient, and the resuscitation targets for each move in opposite directions. There is no algorithm that cleanly resolves that conflict.
My approach in scenarios like this is to treat as though TBI is present when the neurologic examination is unreliable; including intoxication, altered mental status, significant LOC, or if there’s a concerning mechanism.
The hemodynamic risks of more aggressive resuscitation in an actively bleeding patient are real, and different clinicians will weigh those risks differently. What matters is that the competing priorities are explicitly acknowledged and discussed among the treating clinicians (or in your head to yourself); and that the chosen resuscitation strategy reflects a deliberate decision rather than reflexive habit.
The broader theme: resuscitation should be data-informed, individualized, and intentional.
3. Blood Product Strategy in Hemorrhagic Shock
This patient received warmed packed red blood cells. He had a pelvic fractures with posterior presacral venous plexus injury, a right pneumothorax with pulmonary contusions, a right proximal femoral fracture, and an open right tibia fracture. He was exsanguinating, and blood helped to save his life.
The evidence for balanced resuscitation is well-established. Packed red blood cells restore oxygen-carrying capacity but do little to address the coagulopathy that develops rapidly in major hemorrhage. Plasma replaces clotting factors and carries vascular membrane-stabilizing properties beyond factor replacement - protecting and restoring endothelial integrity (Pati et al., 2010). Together, in at least a 1:1 ratio or as whole blood, these components address the primary deficits of hemorrhagic shock simultaneously (Holcomb et al., 2015).
Whole blood is increasingly used in civilian trauma resuscitation and shows promise in prehospital settings (Brown et al., 2025). Cold-stored low-titer O-positive whole blood (LTOWB) offers logistical advantages: one product replaces three and eliminates excess anticoagulant-preservative volume (Brown et al., 2025). Evidence suggests LTOWB may improve survival in patients with elevated probability of mortality and reduce total transfusion requirements, though overall mortality benefits across all trauma patients remain uncertain (Brown et al., 2025). The limiting factors are cost, storage logistics, and shelf life (generally speaking: 21 days for whole blood versus 42 days for PRBCs), which create real barriers for many EMS agencies, particularly ground-based services where implementation remains limited (Brown et al., 2025).
4. Prehospital Tranexamic Acid in Different Trauma Systems
This patient received TXA - a reasonable decision given the high-energy mechanism, clinical hemorrhage, and physical exam findings. The foundational evidence comes from CRASH-2, which found a 1.5% absolute reduction in mortality in bleeding trauma patients when TXA was administered within 3 hours of injury. That effect was time-dependent: benefit was concentrated in patients treated within the first hour (CRASH-2 Trial Collaborators, 2010).
PATCH-Trauma found that prehospital TXA did not translate into better long-term recovery at six months - patients were no more likely to survive with good functional outcomes - despite a signal suggesting fewer deaths when administered in the early phase after injury.
The most recent guidance from NAEMSP, ACEP, and the ACS Committee on Trauma reflects that tension honestly. Their 2025 joint position statement endorses TXA as a reasonable intervention in patients with suspected hemorrhagic shock but stops short of recommending universal prehospital protocols - acknowledging that in systems with rapid access to surgical hemorrhage control, the incremental benefit remains uncertain. It also reinforces two operationally important points: administer within 3 hours of injury (as fast as possible), and communicate administration clearly to receiving teams to prevent duplicate dosing (Barrett et al., 2025).
The practical takeaway: TXA remains a great prehospital intervention for patients with suspected hemorrhagic shock, particularly when given early. If your system carries TXA, administer it early when the clinical scenario supports it - and recognize that getting to definitive care in a timely manner remains the highest-benefit intervention.
5. Analgesia in Hemorrhagic Shock
This patient was yelling in pain, and for some clinicians a 70 mcg dose of fentanyl may feel aggressive in the setting of evolving shock. Untreated pain, however, is not neutral; it drives catecholamine surge, agitation, and impaired assessment. In the immediate post-injury period, trauma patients often enter a hyperdynamic state with enhanced opioid clearance, which may necessitate higher or repeated dosing with close reassessment (Martyn et al., 2019). Moderate opioid doses are generally well tolerated, and national EMS data show that prehospital opioid administration for injured patients - most commonly fentanyl - is associated with meaningful pain reduction and low rates of adverse events (Dalton et al., 2022; De Simone et al., 2024).
The relevant concern is really not the dose necessarily but whether opioid administration could blunt ventilatory drive in a patient who is compensating for metabolic acidosis in the setting of severe trauma, or alter mental status in a way that obscures one of the most important serial markers of perfusion in the field: the patient’s ability to speak, interact, and report clinical change.
This crew managed that balance appropriately. Fentanyl was administered, pain improved, and both mental status and ventilatory effort were reassessed continuously rather than attributing somnolence or respiratory changes reflexively to sedation or intoxication. Some clinicians may prefer rapid, sequential dosing at 0.5 mcg/kg to enhance titratability and reduce the risk of oversedation in a sick, compensated patient. Both approaches are fine in my opinion; I just happen to fall into the higher dose camp.
6. Pelvic Stabilization During Extrication
High-energy pelvic fractures are associated with significant hemorrhage and mortality. Early circumferential pelvic compression decreases intrapelvic volume and tamponades venous bleeding. Commercial pelvic circumferential compression devices (PCCDs) are the recommended level of care (Kuner et al., 2021). In this case, applying one to an entrapped patient with dashboard intrusion was simply not possible.
The crew used the KED during extrication, a practical workaround common in U.S. prehospital practice, though the evidence base supporting this approach is limited. A cadaveric study by Reynard et al. (2016) found that KED use alone had a neutral effect on pelvic movement, whereas the addition of a belt threaded through the thigh straps significantly reduced pelvic mobility. A 2022 regional consensus statement does not formally recommend the KED as a pelvic stabilization device; the rationale is preference for validated PCCDs when available, not that the KED is inherently harmful (New Brunswick Trauma Program, 2022). When a dedicated PCCD cannot be applied to an entrapped patient, use of the KED as a temporary strategy is a great and reliable alternative.
On the question of switching to a dedicated PCCD once the patient reached the stretcher: my personal preference would have been to leave the KED in place if it was providing adequate stabilization. I’m of the mindset that, in significant pelvic injuries, the pelvis should be moved as little as possible. Likewise, the pelvis should be assessed once. Repeated manual palpation of a suspected pelvic fracture risks displacing fragments, causing further injury like vessel lacerations or disrupting a tamponading hematoma (Coccolini et al., 2017).
7. Tension Pneumothorax as a Dynamic Clinical Diagnosis
The right-sided tension pneumothorax evolved during transport. After the patient was moved, vital signs worsened progressively, and repeat auscultation revealed absent right-sided breath sounds in the setting of deteriorating hemodynamics. This is not atypical. In blunt thoracic trauma, tension pneumothorax may develop or worsen over time, and detection can be challenging; auscultation in the field during active resuscitation is inherently difficult in a noisy environment (somewhere, the ultrasound enthusiasts just felt a disturbance in the force).
The clinical findings that drove the decision to decompress were sufficient. The 4th ICS midaxillary line approach is consistent with evidence supporting more reliable access in adults compared to the traditional 2nd ICS midclavicular line, particularly in patients with significant chest wall soft tissue (Inaba et al., 2012; Laan et al., 2016).
This patient had no tracheal deviation and no JVD. Both are commonly taught as cardinal signs of tension pneumothorax. A dual reminder that the classic teaching overstates their reliability, and that tension pneumothorax is a clinical diagnosis with variable and often incomplete physical findings, particularly early in its evolution.
Leigh-Smith and Harris (2005) found tracheal deviation absent in all patients across a series of 108 prehospital needle decompressions. Their conclusion was unambiguous: tracheal deviation and JVD should be de-emphasized, if not abandoned, as taught diagnostic criteria. Both are pre-terminal findings - meaning that by the time they appear, they suggest imminent circulatory collapse.
JVD has an important prerequisite: sufficient circulating blood volume to distend the jugular veins. In the setting of significant hemorrhage, intravascular volume is depleted, and there may simply not be enough circulating blood to produce visible venous distention.
The reliable early clinical findings in an awake patient are respiratory distress, hypotension, tachycardia, tachypnea, falling SpO₂, and unilateral breath sound changes, interpreted in the context of mechanism and chest wall injury.
About finger thoracostomy: My personal recommendation is finger thoracostomy for any patient - prehospital or in-hospital - in whom decompression is indicated in an acute resuscitation. Finger thoracostomy provides definitive pleural access with direct tactile confirmation; when your finger enters the chest cavity, you know you are in. Needle decompression can fail due to inadequate catheter length, kinking, clotting, or chest wall thickness - with reported failure rates in some series exceeding 50%, depending on technique and patient anatomy (Inaba et al., 2012; Laan et al., 2016).
That said, I recognize that most EMS systems operate under protocols where needle decompression remains the standard of care. In those systems, use of an adequate-length commercial catheter at the appropriate landmark represents the best current evidence-based approach.
8. Mode of Transport and Destination Decision-Making
Two transport decisions shaped this call: the choice to go ground rather than air, and the choice to bypass the Level 3 center and transport directly to the Level 1 thirty minutes away.
On the air versus ground question: the literature here is genuinely mixed, which is part of why the decision is harder than it looks. Several large studies and registry analyses - including work from the Pennsylvania Trauma Outcomes Study and National Trauma Data Bank - have found survival benefits associated with helicopter transport, particularly for higher-acuity patients (Brown et al., 2016; Galvagno et al., 2012). Other studies, including analyses of patients with hypovolemic shock and TBI, found no difference in adjusted mortality between air and ground transport when advanced life support was available on the ground (Bulger et al., 2012). Pennsylvania Trauma Registry data show that helicopter transport does not offer a time advantage at shorter distances. HEMS becomes faster than ground transport only when the scene is more than about 7.7 miles from the trauma center; in poor weather, that threshold increases to over 17 miles. At distances closer than that, ground transport is generally faster or equivalent (Chen et al., 2018).
This crew faced that challenge – which is better for the patient? Requesting a helicopter involves rotor spin-up time, identifying and securing a landing zone, transferring care to a flight crew, and moving a critically ill patient mid-resuscitation. Even under ideal conditions, these steps introduce delay and disruption; under less-than-ideal conditions, additional variables such as weather, landing complications, or interval clinical deterioration can further extend time to definitive care.
In this case, the ground crew already had three ALS clinicians in the back, blood product infusing, and a 25- to 30-minute transport time to a Level 1 trauma center.
The question is not simply which mode is faster, but which mode preserves uninterrupted, high-quality resuscitation while expediting arrival at the most appropriate destination. In this case, ground transport was the right decision.
The question of where to take the patient is frequently misapplied in practice. Prehospital trauma triage guidelines in most US systems - adapted from the American College of Surgeons Committee on Trauma Field Triage Guidelines - direct major trauma patients to the highest-level appropriate trauma center. A Level 3 trauma center 13 minutes away is not the appropriate destination for a patient with multisystem trauma, hemodynamic instability, and a mechanism producing injuries that require surgical hemorrhage control. Available literature consistently supports direct transport to a Level 1 or Level 2 center for severely injured patients, with secondary interfacility transfer associated with increased mortality when compared to primary direct transport (Deeb et al., 2023; Gough et al., 2020). The crew chose correctly. The additional 17 minutes of transport time was worth it.
The broader principle: a paramedic crew providing damage-control resuscitation in the back of an ambulance is providing mobile intensive care. The patient is not waiting at a Level 3 for anything that the EMS crew cannot deliver en route to the Level 1. He is waiting for a trauma surgeon and an OR.
The Learning Points
1. A Structured Shock Bundle
My approach is to use a structured “shock bundle” for assessment in the prehospital or austere environment. Although not a formally validated composite score, it intentionally integrates complementary, readily available physiologic data. Shock index, ETCO₂, mental status, and skin perfusion (capillary refill) together create a dynamic framework that helps clinicians identify early deterioration and anticipate decompensation rather than react to it.
Shock index should be calculated with every set of vital signs in any acutely ill patient, not just trauma. In my experience, it is a simple, high-yield tool for identifying potential instability. For example, in the emergency department, when triaging a long list of new patients, I mentally prioritize them using a combination of chief complaint, ECG findings, and shock index - my own informal modification of the ESI system.
2. Blood > Crystalloid
When blood is available, use it. PRBCs restore oxygen-carrying capacity, but balanced resuscitation is the goal. Blood addresses the primary deficits of hemorrhagic shock; crystalloid does not. When blood is not immediately available, small aliquots of isotonic crystalloid titrated to permissive hypotension targets can serve as a bridge, but they are not ideal. Avoid reflexive large-volume boluses in suspected acute hemorrhage. If your system does not carry blood, that is a medical director conversation worth having.
3. Analgesia Requires Vigilant Reassessment
Pain control is not optional in trauma care. In the setting of hemorrhagic shock, the risk of opioids lies less in the dose itself and more in the potential to obscure two critical data streams: mental status and ventilatory effort. Untreated pain worsens catecholamine surge, agitation, and assessment quality and thus ability for you to identify potential life-threatening injuries. Give the medication. Reassess continuously. Treat any somnolence or respiratory alteration as potential shock until proven otherwise.
4. Early Pelvic Stabilization
Suspected pelvic fracture warrants one careful assessment. Repeated palpation risks further displacement and vascular injury. Apply circumferential compression as early as the clinical situation allows and leave a properly positioned device in place.
5. Tension Pneumothorax Is a Clinical Diagnosis
Tracheal deviation and JVD are teaching constructs and often late findings. In blunt thoracic trauma with appropriate mechanism, chest wall injury, unilateral absent breath sounds, progressive hypoxia, and hemodynamic deterioration, that is your diagnosis. Do not wait for textbook signs that may never appear.
6. Take the Right Patient to the Right Place, by the Right Mode
Direct transport to the highest-level appropriate trauma center improves outcomes in major trauma. Secondary interfacility transfer costs time and is associated with worse outcomes. Air transport is not automatically superior; continuity of high-quality resuscitation matters.
Prehospital Case Review is intended for educational purposes only and does not constitute medical advice. Clinical decisions should always be made in accordance with your agency’s protocols and under the guidance of your medical director.
Works Cited
Barrett, W. J., Kaucher, K. A., Orpet, R. E., Campion, E. M., Goodloe, J. M., Fischer, P. E., Colwell, C. B., & Lyng, J. W. (2025). Tranexamic acid in trauma: A joint position statement and resource document of NAEMSP, ACEP, and ACS-COT. The journal of trauma and acute care surgery, 99(3), 357–363. https://doi.org/10.1097/TA.0000000000004727
Brown, J. B., Yazer, M. H., Kelly, J., Spinella, P. C., DeMaio, V., Fisher, A. D., Cap, A. P., Winckler, C. J., Beltran, G., Martin-Gill, C., & Guyette, F. X. (2025). Prehospital Trauma Compendium: Transfusion of Blood Products in Trauma - A Position Statement and Resource Document of NAEMSP. Prehospital emergency care, 1–10. Advance online publication. https://doi.org/10.1080/10903127.2025.2476195
Brown, J. B., Gestring, M. L., Guyette, F. X., Rosengart, M. R., Stassen, N. A., Forsythe, R. M., Billiar, T. R., Peitzman, A. B., & Sperry, J. L. (2016). Helicopter transport improves survival following injury in the absence of a time-saving advantage. Surgery, 159(3), 947–959. https://doi.org/10.1016/j.surg.2015.09.015
Bulger, E. M., Guffey, D., Guyette, F. X., MacDonald, R. D., Brasel, K., Kerby, J. D., Minei, J. P., Warden, C., Rizoli, S., Morrison, L. J., Nichol, G., & Resuscitation Outcomes Consortium Investigators (2012). Impact of prehospital mode of transport after severe injury: a multicenter evaluation from the Resuscitation Outcomes Consortium. The Journal of Trauma and Acute Care Surgery, 72(3), 567–573. https://doi.org/10.1097/TA.0b013e31824baddf
Bryant, M. K., Portelli Tremont, J. N., Patel, Z., Cook, N., Udekwu, P., Reid, T., Maine, R. G., & Moore, S. M. (2021). Low initial pre-hospital end-tidal carbon dioxide predicts inferior clinical outcomes in trauma patients. Injury, 52(9), 2502–2507. https://doi.org/10.1016/j.injury.2021.07.019
Campion, E. M., Cralley, A., Robinson, C., Sauaia, A., Pieracci, F. M., Lawless, R. A., Platnick, B., Robinson, J., Edwards, D., McVaney, K., Moore, E. E., Cohen, M. J., Coleman, J. J., Fox, C., & Burlew, C. C. (2020). Prehospital end-tidal carbon dioxide predicts massive transfusion and death following trauma. Journal of Trauma and Acute Care Surgery, 89(4), 703–707. https://doi.org/10.1097/TA.0000000000002846
Campion, E. M., Cralley, A., Sauaia, A., Buchheit, R. C., Brown, A. T., Spalding, M. C., & Burlew, C. C. (2022). Prehospital end-tidal carbon dioxide is predictive of death and massive transfusion in injured patients: An Eastern Association for Surgery of Trauma multicenter trial. Journal of Trauma and Acute Care Surgery, 92(2), 355–361. https://doi.org/10.1097/TA.0000000000003447
Cannon, C. M., Braxton, C. C., Kling-Smith, M., Mahnken, J. D., Carlton, E., & Moncure, M. (2009). Utility of the shock index in predicting mortality in traumatically injured patients. Journal of Trauma, 67(6), 1426–1430. https://doi.org/10.1097/TA.0b013e3181bbf728
Carsetti, A., Antolini, R., Casarotta, E., Damiani, E., Gasparri, F., Marini, B., Adrario, E., & Donati, A. (2023). Shock index as predictor of massive transfusion and mortality in patients with trauma: A systematic review and meta-analysis. Critical Care, 27(1), 85. https://doi.org/10.1186/s13054-023-04386-w
Chen, X., Gestring, M. L., Rosengart, M. R., Peitzman, A. B., Billiar, T. R., Sperry, J. L., & Brown, J. B. (2018). Logistics of air medical transport: When and where does helicopter transport reduce prehospital time for trauma? Journal of Trauma and Acute Care Surgery, 85(1), 174–181. https://doi.org/10.1097/TA.0000000000001935
Coccolini, F., Stahel, P. F., Montori, G., Biffl, W., Horer, T. M., Catena, F., Kluger, Y., Moore, E. E., Peitzman, A. B., Ivatury, R., DeAngelis, N., Drucker, W. R., Leppaniemi, A., Manfredi, R., Magnone, S., Goretta, O., & Ansaloni, L. (2017). Pelvic trauma: WSES classification and guidelines. World Journal of Emergency Surgery, 12, 5. https://doi.org/10.1186/s13017-017-0117-6
CRASH-2 Trial Collaborators. (2010). Effects of tranexamic acid on death, vascular occlusive events, and blood transfusion in trauma patients with significant haemorrhage (CRASH-2): A randomised, placebo-controlled trial. The Lancet, 376(9734), 23–32. https://doi.org/10.1016/S0140-6736(10)60835-5
Dalton, M. K., Semco, R. S., Ordoobadi, A. J., & Hashmi, Z. G. (2022). Opioid administration in the prehospital setting for patients sustaining traumatic injuries: An evaluation of national emergency medical services data. Injury, 53(9), 2923–2929. https://doi.org/10.1016/j.injury.2022.03.068
Deeb, A. P., Teng, C. Y., Peitzman, A. B., Sperry, J. L., Brown, J. B., & Guyette, F. X. (2023). Direct trauma center access by helicopter emergency medical services is associated with improved survival after severe injury. Annals of Surgery, 278(4), e840-e847. https://doi.org/10.1097/SLA.0000000000005812
De Simone, B., Chouillard, E., Podda, M., et al. (2024). The 2023 WSES guidelines on the management of trauma in elderly and frail patients. World Journal of Emergency Surgery, 19(1), 18. https://doi.org/10.1186/s13017-024-00537-8
Galvagno, S. M., Haut, E. R., Zafar, S. N., Millin, M. G., Efron, D. T., Koenig, G. J., Baker, S. P., Bowman, S. M., Pronovost, P. J., & Haider, A. H. (2012). Association between helicopter vs ground emergency medical services and survival for adults with major trauma. JAMA, 307(15), 1602–1610. https://doi.org/10.1001/jama.2012.467
Gough, B. L., Painter, M. D., Hoffman, A. L., Pritts, T. A., Goodman, M. D., Campion, E. M., Guillamondegui, O. D., & Stassen, N. A. (2020). Right patient, right place, right time: Field triage and transfer to level I trauma centers. The American Surgeon, 86(5), 400-406. https://doi.org/10.1177/0003134820918249
Holcomb, J. B., Tilley, B. C., Baraniuk, S., Fox, E. E., Wade, C. E., Podbielski, J. M., del Junco, D. J., Brasel, K. J., Bulger, E. M., Callcut, R. A., Cohen, M. J., Cotton, B. A., Fabian, T. C., Inaba, K., Kerby, J. D., Muskat, P., O’Keeffe, T., Rizoli, S., Robinson, B. R. H., & PROPPR Study Group. (2015). Transfusion of plasma, platelets, and red blood cells in a 1:1:1 vs a 1:1:2 ratio and mortality in patients with severe trauma: The PROPPR randomized clinical trial. JAMA, 313(5), 471–482. https://doi.org/10.1001/jama.2015.12
Inaba, K., Ives, C., McClure, K., Branco, B. C., Eckstein, M., Shatz, D., Martin, M. J., Reddy, S., & Demetriades, D. (2012). Radiologic evaluation of alternative sites for needle decompression of tension pneumothorax. Archives of surgery (Chicago, Ill. : 1960), 147(9), 813–818. https://doi.org/10.1001/archsurg.2012.751
Kuner, V., van Veelen, N., Studer, S., Van de Wall, B., Fornaro, J., Stickel, M., Knobe, M., Babst, R., Beeres, F. J. P., & Link, B. C. (2021). Application of Pelvic Circumferential Compression Devices in Pelvic Ring Fractures-Are Guidelines Followed in Daily Practice?. Journal of clinical medicine, 10(6), 1297. https://doi.org/10.3390/jcm10061297
Laan, D. V., Vu, T. D., Thiels, C. A., Pandian, T. K., Schiller, H. J., Murad, M. H., & Aho, J. M. (2016). Chest wall thickness and decompression failure: A systematic review and meta-analysis comparing anatomic locations in needle thoracostomy. Injury, 47(4), 797–804. https://doi.org/10.1016/j.injury.2015.11.045
Leigh-Smith, S., & Harris, T. (2005). Tension pneumothorax - time for a re-think? Emergency Medicine Journal, 22(1), 8–16. https://doi.org/10.1136/emj.2003.010421
Levy, M. J., Schaefer, R. M., Obyrne, H., Krohmer, J. R., Bank, E. A., & Holcomb, J. B. (2025). Prehospital blood transfusion coalition clinical practice guideline for civilian emergency medical services. Trauma Surgery & Acute Care Open, 10, e001931. https://doi.org/10.1136/tsaco-2025-001931
Martyn, J. A. J., Mao, J., & Bittner, E. A. (2019). Opioid tolerance in critical illness. New England Journal of Medicine, 380(4), 365-378. https://doi.org/10.1056/NEJMra1800222
McMullan, J., Curry, B. W., Calhoun, D., Forde, F., Gray, J. J., Lardaro, T., Larrimore, A., LeBlanc, D., Li, J., Morgan, S., Neth, M., Sams, W., & Lyng, J. (2024). Prehospital Trauma Compendium: Fluid resuscitation in trauma - a position statement and resource document of NAEMSP. Prehospital Emergency Care. Advance online publication. https://doi.org/10.1080/10903127.2024.2433146
New Brunswick Trauma Program. (2022). Consensus statement: Pelvic injury management. Horizon Health Network / Vitalite Health Network. https://nbtrauma.ca/wp-content/uploads/2022/08/FINAL-Pelvic-Injury-Management-and-appendices-August-2022-EN.pdf
PATCH-Trauma Investigators and the ANZICS Clinical Trials Group, Gruen, R. L., Mitra, B., Bernard, S. A., Burns, B., Smith, K., Cooper, D. J., Gantner, D., Higgins, A. M., Stephenson, M., Myles, P. S., Bellomo, R., & Murray, L. (2023). Prehospital tranexamic acid for severe trauma. New England Journal of Medicine, 389(2), 127–136. https://doi.org/10.1056/NEJMoa2215457
Pati, S., Matijevic, N., Doursout, M. F., Ko, T., Cao, Y., Deng, X., Kozar, R. A., Hartwell, E., Conyers, J., & Holcomb, J. B. (2010). Protective effects of fresh frozen plasma on vascular endothelial permeability, coagulation, and resuscitation after hemorrhagic shock are time dependent and diminish between days 0 and 5 after thaw. Journal of Trauma, 69(Suppl 1), S55–S63. https://doi.org/10.1097/TA.0b013e3181e453d4
Reynard, F., Flaris, A. N., Simms, E. R., Rouviere, O., Roy, P., Bouzat, P., & Payen, J. F. (2016). Kendrick extrication device and unstable pelvic fractures: Should a trochanteric belt be added? A cadaveric study. Injury, 47(3), 711–716. https://doi.org/10.1016/j.injury.2016.01.028